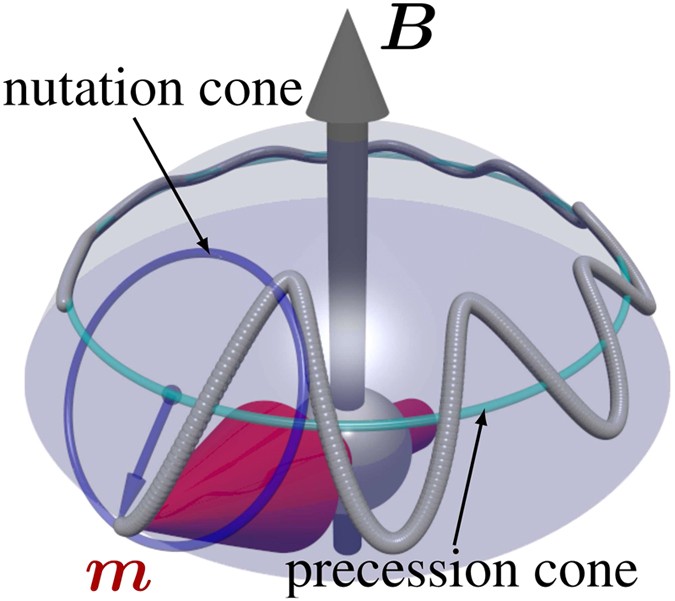
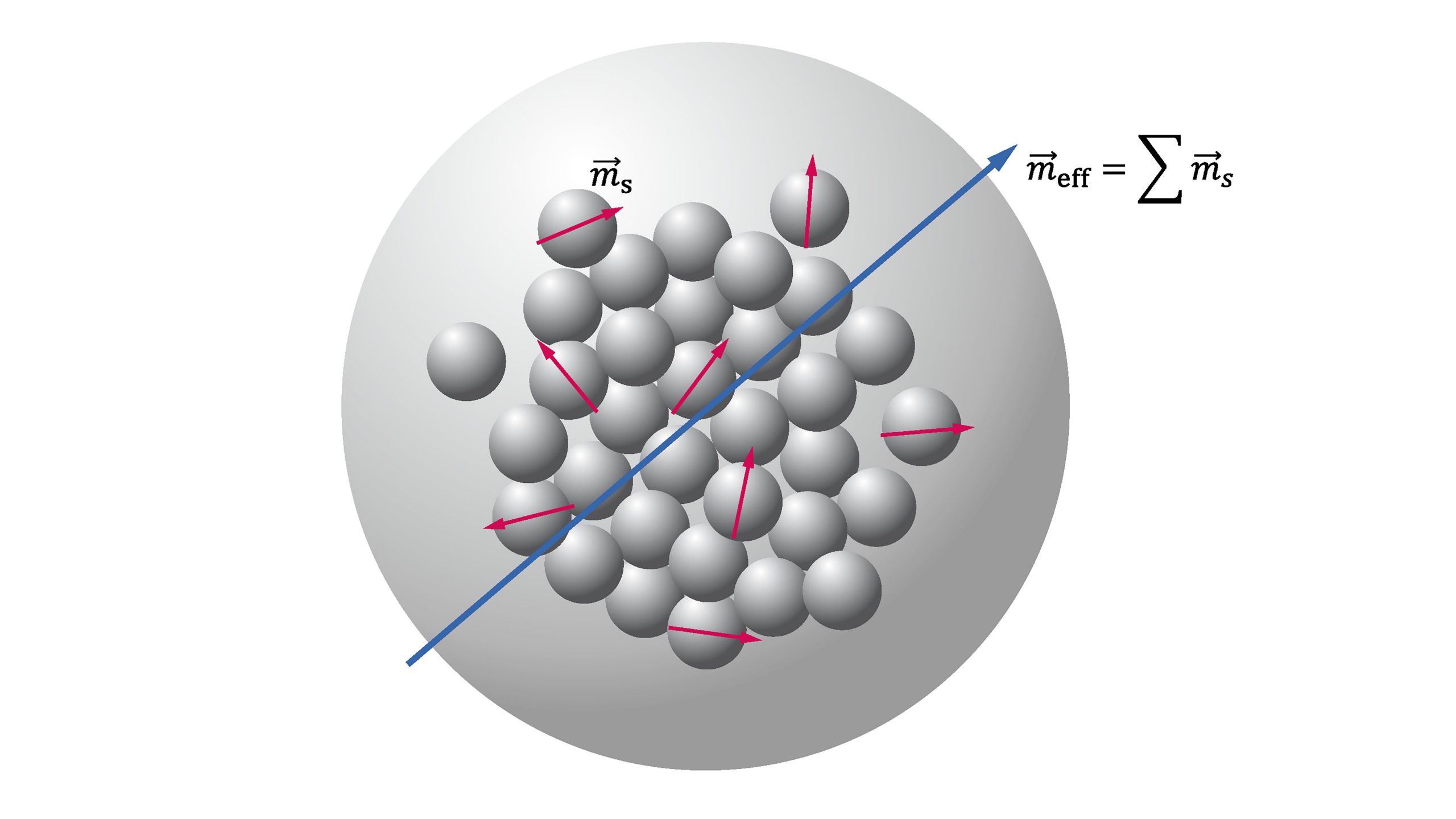
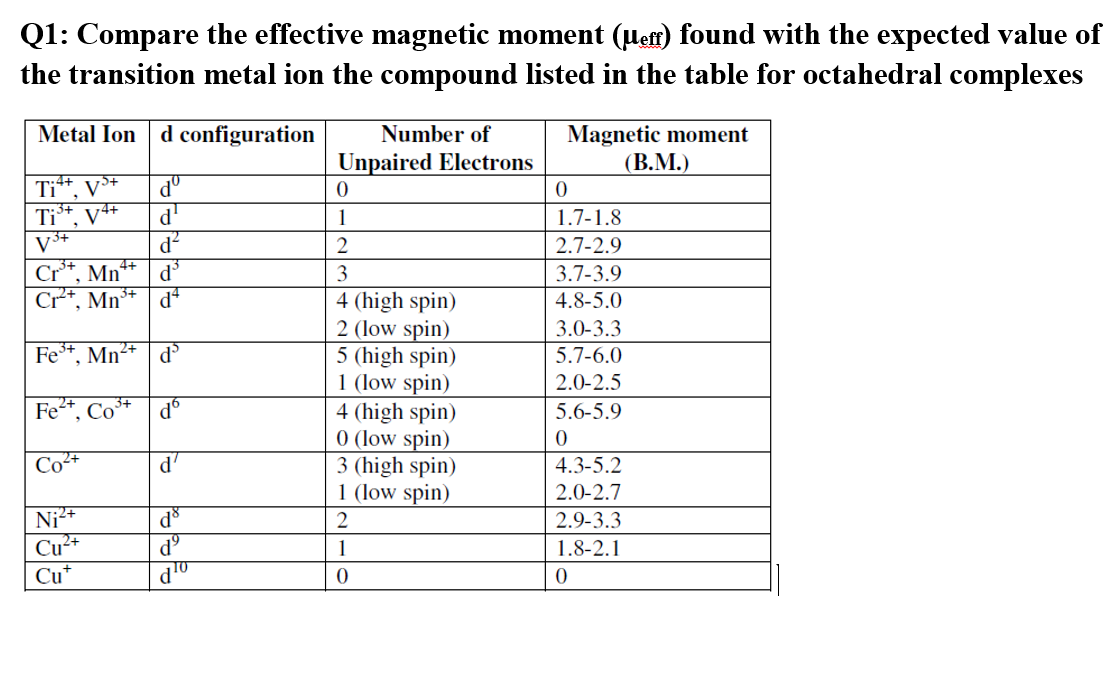
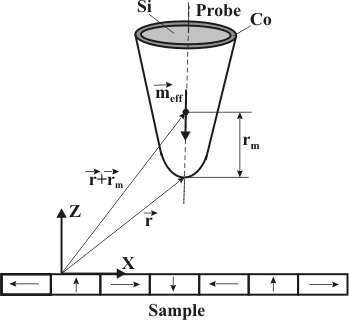
The effective magnetic moments of Co2+ and Co3+ in SrTiO3 investigated by temperature-dependent magnetic susceptibility - ScienceDirect

homework and exercises - Calculating the magnetic field of the Proton in Spin-orbit Coupling in Hydrogen Atom - Physics Stack Exchange

The effective magnetic moment of the system shown in the figure is: (p is the pole strength and 2a is the length of each side of the triangle) -22 SO2a/ a) 2ap